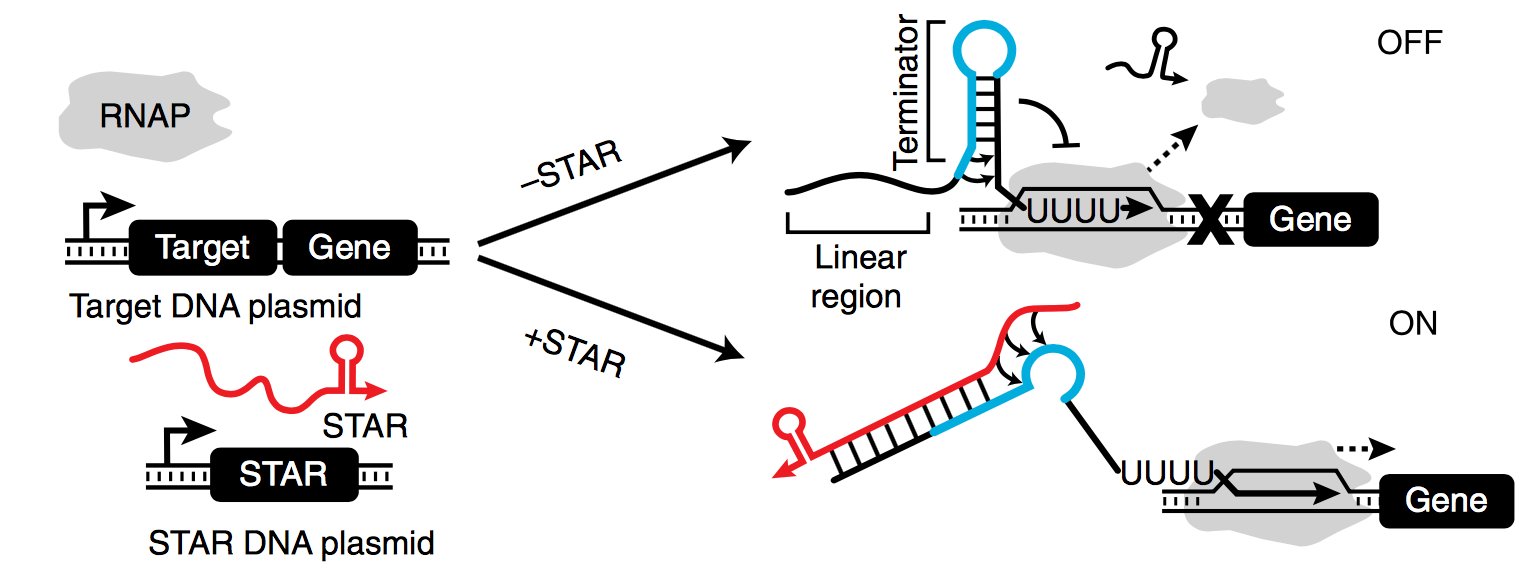
Research led by a former Lucks Lab postdoc, Dr. James Chappell (now an Assistant Professor in BioSciences at Rice University), was recently published in Nature Communications. The research article “Computational design of small transcription activating RNAs for versatile and dynamic gene regulation” describes the lab’s latest work on a novel and synthetic mechanism for the regulation of gene expression. Small Transcription Activating RNAs (STARs), first described in Nature Chemical Biology, represent a completely synthetic mode of gene regulation at the transcriptional level in which an intrinsic terminator hairpin is placed upstream of the gene to be regulated. In the absence of a STAR, formation of the terminator hairpin prevents gene expression by halting transcription. In the presence of the STAR, the terminator hairpin is disrupted and transcription is allowed to proceed.

The latest STARs paper describes a computational approach that leverages computational RNA structure design tools to create a large library of STARs, with the largest dynamic range ever observed for an RNA-based mechanism (>9,000-fold activation). Furthermore, these next-generation STARs are highly orthogonal and can regulate expression in diverse contexts, both in vivo and in cell free extracts, to tune and optimize multigene metabolic pathways and endogenous E. coli genes, and expand RNA networks.
Matthew Verosloff, a PhD candidate in Northwestern’s Interdisciplinary Biological Sciences Graduate Program who was involved in the work, describes the major advancement of this work as improving “not only the regulation capabilities of pathways to virtually on or off,” and building “a large and diverse toolset with tremendous orthogonality between parts.”
To read more about the work, check out Northwestern’s news article “Creating a Better RNA Switch.”